Want to compromise brain activity? Just add SARS-CoV-2 spike!
Glia-glia, neuron-neuron and neuron-glia fusion to compromise neuronal activity
This latest paper published in Science Advances on June 7, 2023 entitled: “SARS-CoV-2 infection and viral fusogens cause neuronal and glial fusion that compromises neuronal activity”,1 confirms some disturbing theories as to why we have been witnessing neurological impairment and damages since SARS-CoV-2 was introduced to the public. It also begs some extremely disturbing questions as to why the injection-associated spike may also be causing neurological adverse events that are being excessively reported in all pharmacovigilance databases all over the world. Could it be due to the same mechanism?
I made a video about 2 years ago (and testified at the National Citizen’s Inquiry where this is also discussed), where I propose that free (circulating) spike protein2 can bind and disrupt the closed loop cycle of the Renin Angiotensin Aldosterone System (RAAS) by binding ACE-2 to inhibit its activity via competitive binding. This would also have consequences for downstream signaling events in the case of binding membrane-bound ACE-2.
I have also hypothesized that the injection-associated spike protein may be capable of binding ACE-2 in its translated conformation to allow downstream intracellular signaling events to ensue in the cell. This is pure speculation, but bear with me. It has long been the claim of the manufacturers of the injection-associated spike template that the spike protein, once translated by the host’s cellular ribosomes, would fold/conform to a closed pre-fusion state (Figure 1 - second structure) due to two proline mutations (called spike 2P or S-2P, for short short), which in effect, would stabilize the protein and theoretically disallow bindability to ACE-2 due to being locked into this ‘closed’ pre-fusion state. One of the things that bothers me about this is that in the pre-fusion state, the RBD can be in the up state and is ~55% of the time (Figure 1 - third structure) according to Ke et al.3 (N.B. this is in the context of in-tact virions).

The authors of the Nature paper entitled: “Structures and distributions of SARS-CoV-2 spike proteins on intact virions” write:
On the other hand, in the closed conformation of the pre-fusion isoform, all the three RBDs are covered by the N-terminal domain (NTD) of the spike. Ke et al. found that 31% of the pre-fusion spike trimers are closed, 55% have one RBD open, while 14% have two RBD available.4
Just to be clear here, these guys used cryo-EM to map out the structure and distribution of spike on a virion surface. It’s very different from the injection-associated spike situation. It’s very cool and shows a ‘preferred’ pre-fusion ‘native’ state which is not the closed conformation.
Here’s another nagging bit of speculation. What if the terminology ‘pre-fusion state’ has been maligned like the term vaccine has been maligned? I am skeptical of absolutely everything I have read and been told on this subject matter, hence my speculation. Based on the fact that we have not been allowed to sequence the injection-associated spike template, how can we actually know that the 2 proline mutations are there in the first place? Should we simply go on good faith? My stance at this point is: Show me the mutations in many repeat experiments sampling many, many vials, and then I’ll believe it. There are compelling evidences of neurological harms in the context of the injection-associated spike protein as per the millions of adverse event reports in pharmacovigilance databases, including VAERS. I will return to this.
In addition to the potential danger of spike RBD:ACE-2 binding (either the free spike, or hypothetical injection-associated spike) to induce downstream signaling, there is also the danger that arises from the spike’s ability to induce fusion and to potentially disrupt cellular functioning and activity. That’s what this newest paper addresses in the context of brain and central nervous system cells in the context of SARS-CoV-2.
Questions to think about:
Is the injection-associated spike fusogenic?5
Is the difference between individuals who suffer serious neurological consequences and those who do not, simply due to the percentage integrity of the injected RNA template to produce fusogenic spike?
In their methodologies, the authors use mammalian neurons in culture, C. elegans as an in vivo system, mice as a mammalian model system with human ACE-2 (hACE-2) and human neurons, to determine whether or not certain types of brain cells fuse in the presence of SARS-CoV-2 in these specific contexts. They use good controls. They also have absolutely stunning high resolution images and videos as part of their results. Just saying. Really easy on the eyes.
The authors write:
We show that SARS-CoV-2 infection induces fusion between neurons and between neurons and glia in mouse and human brain organoids. We reveal that this is caused by the viral fusogen, as it is fully mimicked by the expression of the SARS-CoV-2 spike (S) protein.
…increasing evidence has revealed the presence of viral RNA and proteins also in the brain and a multitude of neuropsychiatric syndromes.
…hACE2 is expressed in neuronal and glial cells in the human central nervous system.
Let’s back up a bit. What is a fusogen?
On fusogens
In a few words, fusogens are membrane-embedded proteins that mediate cell-cell or virus-cell fusion. In the case of a virus-cell fusion, the proteins on the surface of a virus allow the virus to fuse with the membrane of a cell it is ‘trying to infect’ in order to dump its genetic material and other stuff in the cytosol of the cell to maintain its ‘productive’ cycle. That was a terrible sentence.
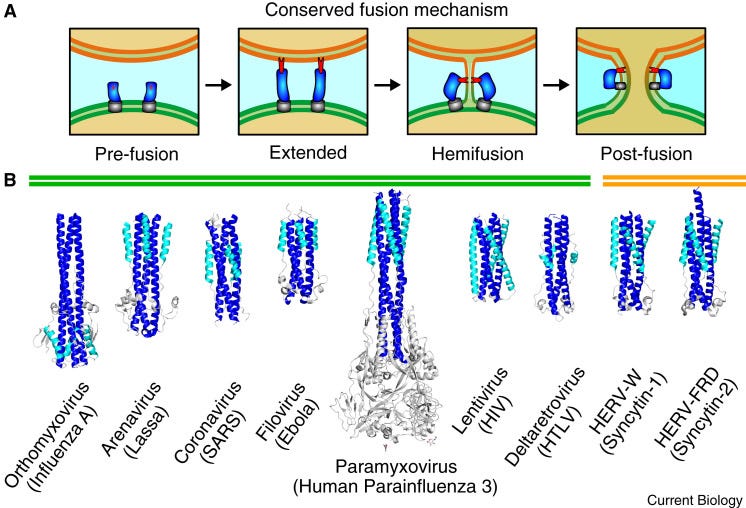
Fusion of membranes is not just good for viruses to infect cells though.
The fusion of two membrane bilayers is critical to eukaryotic life, occurring in gamete fusion, muscle differentiation, ocular lens formation, neurotransmitter release, and placenta formation. Ironically, it is through a similar process that enveloped viruses gain access to eukaryotic cells, merging membranes to release viral genetic material within the cytoplasm. In all cases, membrane fusion is exceptionally unfavorable without the assistance of specialized proteins to overcome the repulsive forces between membranes.6
N.B. It is always important not to demonize elements and members of nature - even retroviruses. For example, without a particular human endogenous retrovirus (HERV), we wouldn’t be able to reproduce.789 Oh, and we are (genomically) ~8% retrovirus.10
The expression of HERV-K, a biologically active family of HERV, produces proteins found in placenta. Furthermore, the expression of the envelope genes of HERV-W (ERVW-1) and HERV-FRD (ERVFRD-1) produces syncytins which are important for the generation of the syncytiotrophoblast cell layer during placentogenesis by inducing cell-cell fusion.11
So fusogens are important in many contexts, and in the context of SARS-CoV-2, the S2 portion is the means by which the virus eventually dumps its genetic material into host cells. Once the trimeric version of the spike is cleaved by TMPRSS2, the S2 portion undergoes a conformational change to allow it to penetrate the host cell membrane and then, kind of like a winch, the S2 folds back on itself and winches the membranes together, even in the face of the repulsive forces.
On glial cells
Glia, also called glial cells (gliocytes) or neuroglia, are non-neuronal cells in the central nervous system (brain and spinal cord) and the peripheral nervous system that do not produce electrical impulses.12
One particular type of glial cell is called an oligodendrocyte. These cells are responsible for the myelination of the axons of neurons, in addition to Schwann cells. So when you hear about demyelinating diseases such as Multiple Sclerosis, think destruction of oligodendrocytes. Oligodendrocytes are located in the central nervous system whereas Schwann cells are located in the peripheral nervous system. These two myelinating cell types function in the same way, just in different locations.
On neural cells/neurons
Within a nervous system, a neuron, neurone, or nerve cell is an electrically excitable cell that fires electric signals called action potentials across a neural network.13
So the neurons convey the signals or messages. In Figure 3, the neuron is the yellow long cell. The myelin coating increases conductivity by increasing the rate of propagation of action potentials along the axon of the neuron. See: Nodes of Ranvier. Not to be confused with the metal band.
So the many different types glial cells have many functions like protecting the neurons by surrounding them, holding them in place, feeding them oxygen and nutrients, insulating them and getting rid of them when they die, and can be micro and macro and localized to the central or peripheral nervous system.
Think of human myelinating glia as being a snow suit, if you live in the North. It would be easy to understand that problems might ensue, and what kind of problems, if the snow suit fused to your skin, or if the snow suit fused to someone else’s snowsuit. How would you poop, for example?
The authors, in their thoroughness, tested the fusogenicity (the ability to induce fusion) in the context of SARS-CoV-2, the p15 fusogen isolated from the baboon orthoreovirus, and the spike protein from SARS-CoV-2 - both in the context of binding ACE-2 and not. They examined the latter by testing to fusogenicity in the context of both the S-2P and the S-6P versions of spike.
On Results
They observed fusion of neural cells in the contexts of SARS-CoV-2, p15 alone and spike protein when bound to ACE-2. They did not observe fusion of neural cells in the context of either of the S-2P or S-6P spike conformations, and this was based on the idea that they can’t bind ACE-2. Both non-2P spike and ACE-2 are required in order to induce fusion of neural cells via binding.

You can see in G, that neither of the proline-mutated versions of spike S induced neuronal fusion. The authors did make these mutations themselves14, and although this result should make me feel better, it doesn’t. Both the p15 and spike:ACE-2 induced neuronal fusion as shown in Figure 5.
With both p15 and spike S, we observed not only neuron-neuron fusion but also neuron-glia and glia-glia fusion when the fusogens were expressed in these cell types (fig. S4), a result that mimics our observation with SARS-CoV-2 infection.
N.B. Skipping to the experiments using human neurons…
On control for human neuron tests
When they tested the fusogenicity of human neurons using two different human embryonic stem cells (hESC)-derived neuronal systems (cortical neurons differentiated in 2D cultures and 3D brain organoids), they chose to use/test the S-6P version of the closed confirmation spike and not the S-2P version. Why? Why not use both or only S-2P? I plan to write to the authors to find out. It seems to me that considering that this is the highest level test they did (ie: using human brain cells - I mean, it’s kind of like one level below human in vivo studies), then they should have used the S-2P mutant so that they could mimic more precisely the potential effects of the spike used in the shots.
On merging contents
They also looked at whether or not the contents of the fused cells were exchanged once fused. And they were. Even mitochondria were shown to be bidirectionally mobile. They also found that if they left the experimental system alone for a few days, then ‘syncytia’ grew to encase/encompass surrounding neural cells. It does not seem a stretch to assume that fusogens globbing together neurons might impair function of these neurons. They looked at that too.
On impairment of neurons
They found that although the fused neurons remain viable, the neuronal activity in ~10% of cells was completely abrogated, and this was tied to somal (the ‘head’ of the cell containing the nucleus and stuff) fusion. In the other 90%, the activity was synced with regard to calcium spikes. Interestingly, in the case of neurons fusing with glial cells, there was a complete loss in neuronal activity. Think back to not being able to poop in your fused snowsuit.
Discussion
This is very concerning to me. If we consider these results, oligodendrocytes (myelinating cells in the brain) can fuse to neurons in the presence of spike:ACE-2 and thus lose function. Think about that. What if the spike translated from the injection-template is not the S-2P version? I know this is unlikely, but until I see proof that it is not the case, I remain open to the possibility.
Since the S-2P mutant disallows binding with ACE-2 (which it certainly should!), and the injection-associated spike gene contains this mutant, then why are there so many reports of neurological-associated adverse events in VAERS and EUDRA and Yellowcard and DAEN? Novacula Occami surmises that the shaven yak is the one to observe. Having said that, whatever it is I just said, it is possible that these neurological adverse events we are observing in the context of the COVID shots are due to a different mechanism of action. This is highly unlikely, in my opinion, and even if this is the case, this is even more reason to open a plethora of new investigations.
The questions for me remain (excuse the repetition - reflective of my state of mind, I ‘spose): Do the injection spike templates really have the proline mutations and/or do these mutations yield a fusion-inactive version of spike in vivo? I am skeptical. I am skeptical because of this paper’s beautiful demonstration of a mechanism of action of neurological damages in the form of compromised neuronal activity in the context of SARS-CoV-2 via spike as a fusogen.
Reports of neurological impairments and dysfunctions are entirely atypical in comparison to reports made historically in pharmacovigilance databases (See Figure 7 for VAERS example). These databases, such as VAERS, are designed for the sole purpose of detecting safety signals in the context of reported vaccine-associated adverse events, meant to be subsequently analyzed using causality assessments (Bradford Hill Criteria), Proportional Reporting Ratio (PRR) or Bayesian analyses. Currently, in VAERS, there are N = 439,673 reports of neurological-associated adverse events for the Domestic and Foreign data sets and N = 305,431 in the Domestic set alone.15 With an under-reporting factor of 31, that becomes N = 13,629,863 and N = 9,468,361 actual occurrences.

If these neurological adverse event occurrences aren’t the direct the effects of the injection-associated spike, then horror of horrors, it is possible that the people experiencing them are immunocompromised by the shots, and subsequently succumbing to SARS-CoV-2 as a result (this is a thing - COVID-19 remains the number 1 reported adverse event in VAERS as of June 9, 2023), and in turn succumbing to neurological damage due to the fusogenic nature of the spike of the virus?
One and done would have new meaning.
To circle back to the paper, the authors write the following in the Discussion:
Our results indicate that viral infections, driving the expression of viral fusogens, can initiate the irreversible fusion of brain cells, causing alteration in neuronal communication and revealing a possible pathomechanism of neuronal malfunction caused by infection. The impact on neuronal fusion will depend on the viral load in the brain and the specific areas infected; for example, in the case of SARS-CoV-2, neuronal fusion depends on the expression of hACE2 and, potentially, other accessory entry factors such as TMPRSS2 and NRP1 in neighboring neurons.
The authors conclude this article with a warning:
…our findings demonstrate that it will be critical to consider the fusogenic potential when designing any future vaccines in which viral fusogens are to be expressed in mammalian cells.
The damages done in the context of ACE-2-binding, as is the case with SARS-CoV-2, will completely depend on the pattern of ACE2-expressing cells in the host. Unfortunately as previously quoted, hACE2 is expressed in neuronal and glial cells in the human central nervous system.
I leave you with the results from a query of Creutzfeldt-Jakob disease, Alzheimer’s disease, Parkinson’s disease, Amyotrophic lateral sclerosis, Huntington’s disease and Multiple sclerosis reports from VAERS for the past 12 years. The last 2 years (2021-2022) only include reports from the COVID injections while the first 10 years (2011-2020) include reports from all vaccines combined. I omitted 2023 since we’re only half-way through. There are more than twice as many reports made for the COVID products alone, per 100,000 shots, when compared to the past 10 years of adverse event reports for all vaccines combined. That’s not nothing.

Ramón Martínez-Mármol et al., SARS-CoV-2 infection and viral fusogens cause neuronal and glial fusion that compromises neuronal activity.Sci. Adv.9,eadg2248(2023).DOI:10.1126/sciadv.adg2248
Yonker LM, et al., Circulating Spike Protein Detected in Post-COVID-19 mRNA Vaccine Myocarditis. Circulation. 2023 Jan 4. doi: 10.1161/CIRCULATION. AHA.122.061025. Epub ahead of print. PMID: 36597886.
Ke, Z., Oton, J., Qu, K. et al. Structures and distributions of SARS-CoV-2 spike proteins on intact virions. Nature 588, 498–502 (2020). https://doi.org/10.1038/s41586-020-2665-2.
Ismail, A.M., Elfiky, A.A. SARS-CoV-2 spike behavior in situ: a Cryo-EM images for a better understanding of the COVID-19 pandemic. Sig Transduct Target Ther 5, 252 (2020). https://doi.org/10.1038/s41392-020-00365-7.
Hsieh CL, Goldsmith JA, Schaub JM, DiVenere AM, Kuo HC, Javanmardi K, Le KC, Wrapp D, Lee AG, Liu Y, Chou CW, Byrne PO, Hjorth CK, Johnson NV, Ludes-Meyers J, Nguyen AW, Park J, Wang N, Amengor D, Maynard JA, Finkelstein IJ, McLellan JS. Structure-based Design of Prefusion-stabilized SARS-CoV-2 Spikes. bioRxiv [Preprint]. 2020 May 30:2020.05.30.125484. doi: 10.1101/2020.05.30.125484. Update in: Science. 2020 Jul 23;: PMID: 32577660; PMCID: PMC7302215.
Vance TDR, Lee JE. Virus and eukaryote fusogen superfamilies. Curr Biol. 2020 Jul 6;30(13):R750-R754. doi: 10.1016/j.cub.2020.05.029. PMID: 32634411; PMCID: PMC7336913.
https://www.pbs.org/wgbh/nova/article/endogenous-retroviruses/
Durnaoglu, Serpen & Lee, Sun-Kyung & Ahnn, Joohong. (2022). Syncytin, envelope protein of human endogenous retrovirus (HERV): no longer ‘fossil’ in human genome. Animal Cells and Systems. 25. 1-11. 10.1080/19768354.2021.2019109.
Khodosevich K, Lebedev Y, Sverdlov E. Endogenous retroviruses and human evolution. Comp Funct Genomics. 2002;3(6):494-8. doi: 10.1002/cfg.216. PMID: 18629260; PMCID: PMC2448423.
de Parseval N, Lazar V, Casella JF, Benit L, Heidmann T. Survey of human genes of retroviral origin: identification and transcriptome of the genes with coding capacity for complete envelope proteins. J Virol. 2003 Oct;77(19):10414-22. doi: 10.1128/jvi.77.19.10414-10422.2003. PMID: 12970426; PMCID: PMC228468.
https://en.wikipedia.org/wiki/Endogenous_retrovirus
https://en.wikipedia.org/wiki/Glia
https://en.wikipedia.org/wiki/Neuron
D. Wrapp, N. Wang, K. S. Corbett, J. A. Goldsmith, C. L. Hsieh, O. Abiona, B. S. Graham, J. S. McLellan, Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 367, 1260–1263 (2020).
strings_NEURO <- c("Neuro", "neuro", "cephalo", "cephali", "neuritis", "Ageusia", "Agitation", "Aphasia", "Aphonia", "Areflexia", "Hallucination", "aesthesia", "nerv", "Nerv", "tremor", "Tremor", "Limb immobilisation", "Lumbar", "brain", "Brain", "CNS", "Depressed level of consciousness", "Depression", "Disorientation", "Dissociation", "Dissociative disorder", "Disturbance in attention", "aesthesia", "Dyskinesia", "Euphoric mood", "paresis", "paralysis", "Paralysis", "May-Thurner syndrome", "Memory impairment", "Meningioma", "Psychogenic", "Seizure", "seizure", "Pupil fixed", "Sensory", "sensory", "palsy", "Palsy", "neuron", "Neuron", "Phonophobia", "Photopsia", "Sleep disorder", "Slow response to stimuli", "Slow speech", "Sluggishness", "Somnolence", "Speech", "Spinal", "spinal", "Visual", "Staring", "Subarachnoid haemorrhage", "Taste disorder", "Thinking abnormal", "Tinnitus", "Tongue biting", "Tongue movement disturbance", "Tongue spasm", "Vertigo", "electric", "Electric", "White matter lesion", "Mening", "mening", "twitch", "Motor", "electro", "Electro", "plasticity", "nervous", "spastic", "Motion", "motion", "Incoherent", "conduction", "syndrome", "Head")





The amount of work you accomplish is amazing. I can't grasp every piece of it but I do get the point.🌹
Amazing commentary on this topic, Jessica! My sister-in-law was diagnosed with Parkinson’s disease in 2017. She took two jabs and has progressed faster in her disease than I have seen clinically. (I’m an NP who used to work in specialized Geriatrics). Since leaving my job, (involuntarily) my colleagues have seen more Prion disease in the last 2 years than I ever saw in my whole career!! This explains ALOT!