Dose 3 response much like sore thumb when plotting Foreign data reports of myocarditis.
You've all seen the Dose 2 response for myocarditis in the Domestic data set, but check out the Dose 3 response in the Foreign data set!
BREAKING NEWS! I’ve always wanted to write that.
But this is pretty cool for me being the nerd that I am.
Background
For those of you who don’t know, VAERS data can be downloaded from the VAERS website where you can find and download data going back 30 years. You can also find data for 2022 deemed ‘Domestic data’, which is the adverse event data collected from Americans within the U.S. in 2022, and also ‘Foreign data’, which is adverse event data collected from U.S. citizens living abroad and reports made directly to the manufacturers subsequently sent on to VAERS from various countries around the world.1 I just learned from an amazing reader that the SPLTTYPE column indicates both the manufacturer that submitted the report to VAERS and the country from where the report was sent from. I have more work to do! In any case, it is safe to say, these data are IN VAERS: vetted, logged, entered, uploaded, downloadable. And just to remind everyone, VAERS is a government database and it is a Federal offense to submit a false VAERS entry.
Knowingly filing a false VAERS report is a violation of Federal law (18 U.S. Code § 1001) punishable by fine and imprisonment.
The hot news!
I was prompted to plot the Foreign data from VAERS in the context of myocarditis today. Thank you very much to my fellow crow-loving friend Robert F. Kennedy Jr. for ccing me on this email sent by one Dr. Palmer posing a question about the ratios of reports of stand alone adverse events in the Domestic versus Foreign VAERS data sets.
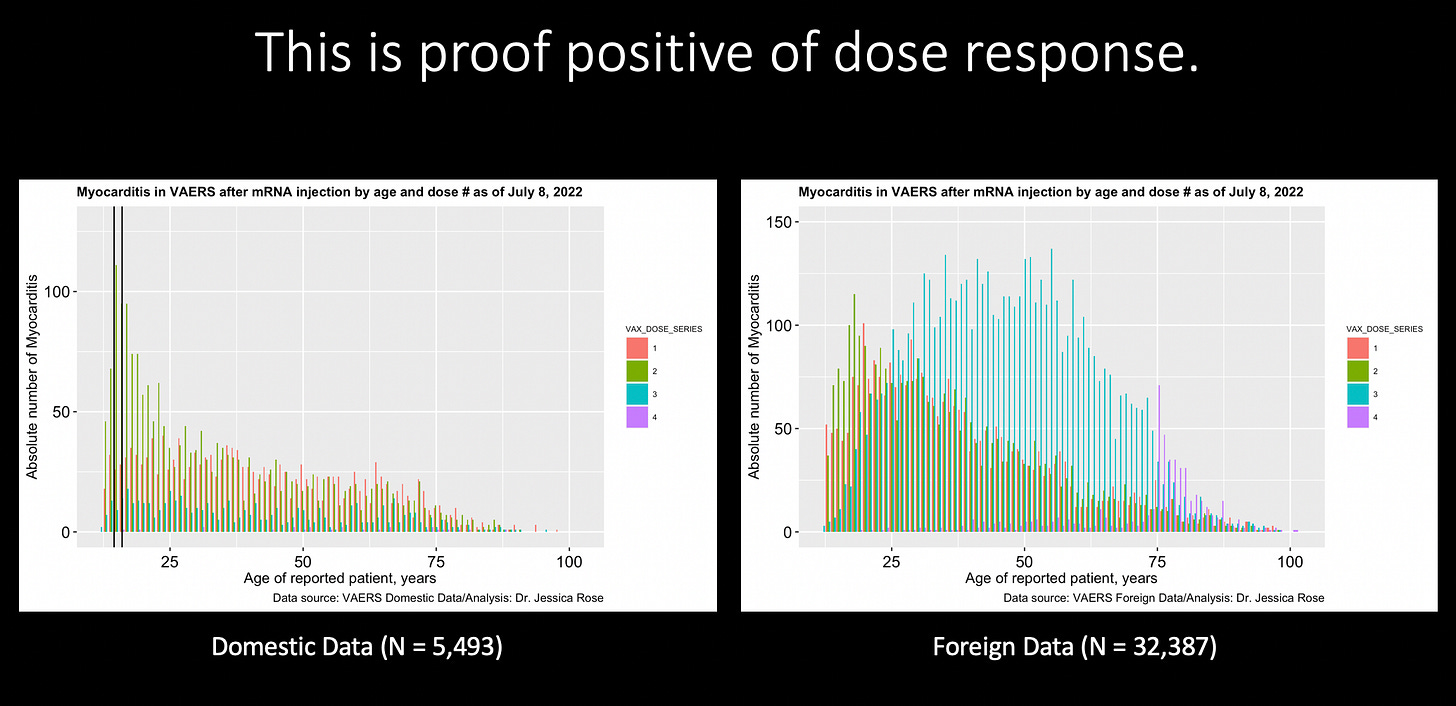
My work on myocarditis (that has gone into publication limbo) showed a dose response effect, primarily in young children. Following Dose 2, there was a mutli-fold greater reporting rate in children age 12-15. The following is Figure 5 from that paper entitled “A Report on Myocarditis Adverse Events in the U.S. Vaccine Adverse Events Reporting System (VAERS) in Association with COVID-19 Injectable Biological Products” (data was up to and including July 9, 2022), and even though the data was sparse then, it still revealed this dose response pattern.

Check out this same plot with the updated VAERS data. By the way, I have been publishing this plot for over a year according to every weekly VAERS update but generally, I do not publish the Foreign data for myocarditis. Just because… I didn’t think to do it! I hadn’t even created this dose response plot for this data set! Until now.

Is this a beautiful thing or what? You don’t often see such incredibly obvious patterns in data - especially data that has been referred to by ‘fact checkers’ as garbage or ‘a dumpster’. This is incredibly satisfying for me. I have been blowing the DOSE RESPONSE horn for over a year now as one of the most important Bradford Hill Criteria to satisfy, that very much is satisfied. Tell me again CDC, how is dose response not satisfied here? When you look at the Foreign reports of myocarditis, it is very clear that Dose 3 is associated with a substantial increase in reporting across most ages (seems up to 75 years).
Interpretation: more shots = mutli-fold increase in reporting rate of myocarditis => more incidents of myocarditis from your shots.
This is what the data looks like when you plot it together (top plot)).

This was very illuminating to me because, I haven’t been plotting the Foreign myocarditis data and therefore didn’t see this obvious Dose 3 response, as I mentioned.
Since I do not know what comprises the population base for the Foreign data, I cannot know how many people were injected with how many doses. I can, however, look at the differences between the Domestic and Foreign data sets with regard to the number of myocarditis reports entered into each. It might not tell me much, but it does beg a serious question that arises from this data:
Why do myocarditis reports comprise 0.67% of the total number of reports of adverse events in the Domestic data set and a staggering 7.6% of the total number of reports of adverse events in the Foreign data set?
The total number of adverse reports in VAERS in the Domestic versus Foreign data sets are not equal to begin with. The Domestic data set contains twice as many total reports as the Foreign: N Domestic = 815,539 vs. N Foreign = 428,261. I only analyze COVID-19-associated reports. So considering the fact that most stand alone adverse events that I have checked are about a 1:1-1.3 ratio with regard to each respective data set (D:F), then why is the split in myocarditis 1:5.9? The only other stand alone adverse event that comes close to this that I have found is Creutzfeldt-Jacob Disease coming in second place with a 1:2.8 ratio. The total numbers of myocarditis reports in VAERS for the Domestic and Foreign data sets are N = 5,493 and N = 32,387, respectively.
The numbers of reports of myocarditis stratified by Dose look like this:
Dose 1 reports - Domestic: N=1,731; Foreign: N=7,948
Dose 2 reports - Domestic: N=2,204; Foreign: N=9,358
Dose 3 reports - Domestic: N=568; Foreign: N=11,618
The reason the Ns don’t add up perfectly to the total Ns is due to missing dose data for each data set: sometimes, the dose data is omitted. But nonetheless, N = 4,503 vs. N = 28,924 is still a 1:6.4 ratio for Domestic vs. Foreign reports.
This article begs another question:
Question: Are particular adverse events being singled out for skewing with regard to Domestic and Foreign data input to hide safety signals?
You be the judge.
This is simply amazing. I just added Dose 4 data.
One more thing: I plotted the total number of reports of myocarditis filed in the Domestic data set in VAERS as per Dose 1, 2 and 3 data normalized to the number of doses administered per dose (People vaccinated, People fully vaccinated and Vaccine booster doses) according to Our World in Data as of July 15, 2022.2 It looks like this.

It is important to confirm the actuality of the dose response and this does. There are truly more reports of myocarditis associated with Dose 2 for the Domestic data. This confirms that the dose response in younger individuals observed by plotting the absolute number of reports is an accurate portrayal of the dose response.
The manufacturer, say Pfizer, has to report all spontaneous reports, even foreign, to VAERS. You'll see that each domestic or foreign report reported to Pfizer (Pfizer then sends to VAERS) has a "Mfr/Imm Project Number." From a reader.
https://ourworldindata.org/covid-vaccinations




David Martin says the jabs will kill 750 million by 2028. Sherri Tenpenny says it will be much more than that a lot sooner. She lists 40 ways these jabs can kill. There will be a tsunami of cancers, brain disorders, heart disease, and A.D.E. in a couple of years. These people who say they got jabbed and feel fine are going to learn an important life lesson: never take medical advice from depopulation fanatics like Gates and Schwab.
Glad to see all the new content. I can’t imagine how exhausting this must be. I have a triple vaxxed friend (52 healthy and super fit). He only eats organic and free range, zero alcohol etc. 5 months after his booster he had an ischemic stroke. He claims it’s due to stress. He also said zero people have died from the vaccine. It’s basically NPR talking points every time. I’ve given up. Glad you’re persisting and making a record. 🤗