Molecular mimicry shown between Multiple Sclerosis-associated proteins and SARS-2 nucleocapsid
They found overlap of 22 MS-associated proteins!
A paper is hot off the presses, published in Scientific Reports 2 days ago (Jan 8, 2022) entitled: “Sequence similarity between SARS-CoV-2 nucleocapsid and multiple sclerosis-associated proteins provides insight into viral neuropathogenesis following infection”.1 Thanks again to David Wiseman for putting this in my email inbox.
I cannot say that I am surprised by this, but people need to know that is paper defines a mechanism of action of ‘a prominent pattern of central nervous system (CNS) pathologies, including demyelination’ defined by Multiple Sclerosis (MS) in the context of SARS-CoV-2. This mechanism of action is molecular mimicry (of CNS proteins).
Now, it is very important to remember here that these homologous sequences they discovered are for the nucleocapsid (N) protein (and replicase polyprotein1ab2) of the SARS-CoV-2 virus, so if the template for the COVID-19 mRNA injectable products encodes the spike protein alone, then people can rest assured they won’t have molecular mimicry insults due to shared epitopes from the N protein to CNS proteins. At least, based on this paper’s results.
MS involves demyelination of nerve cells of the brain and spinal cord and is thought to be the result of immune system dysfunction induced by viral infections, gut dysbiosis, enhanced gut permeability or epigenetic triggers - all which can lead to autoimmunity against the myelin sheath and central nervous system (CNS) proteins.345 Molecular mimicry is implicated in the development of autoimmune diseases67 and is linked to vaccines as well and thus, this is very important to think about - especially these days.8 I hadn’t realized that the mechanistic role of molecular mimicry in autoimmunity was a point of contention, but apparently, it is.
MS is quite common in the U.S. and affects ~3M people world-wide.9 The authors in this recently published paper appear to have identified shared sequences between the SARS-CoV-2 N protein and CNS proteins that that can result in MS manifestation post SARS-CoV-2 exposure. They used a robust new computational tool called PEPmatch10 to assess homology and demonstrated that SARS-CoV-2 may be associated with the development of MS via molecular mimicry of CNS proteins. (I am currently checking this PEPmatch bioinformatics tool out. Apparently a manuscript is in progress. You can access the Github here.)
The authors write:
Central to the theory of molecular mimicry is the degenerative nature of both T cells and MHC binding, in which multiple peptides can bind to the same MHC, and in turn multiple peptide:MHC combinations can be recognized by the same T cell Receptor (TCR).
This so-called degeneration of T cells is driven by an induced ‘flexibility’ of T cells (their receptors) to recognize more than one antigenic epitope.11 Kind of like the Blanche Devereaux’s of the T cell receptor world - not so choosy in their ‘binding partner’.
I have written about molecular mimicry in the context of SARS-CoV-2 previously and you can read about that here. So, I won’t go into detail on it now, but instead refer everyone to the following schematic that describes molecular mimicry as the causative mechanism for autoimmunity in rheumatic heart disease.12 Even though the guys at Johns Hopkins have this blurb and schematic on their site, they still seem skeptical about molecular mimicry being behind autoimmunity. In any case, thanks guys. It’s a nice schematic.
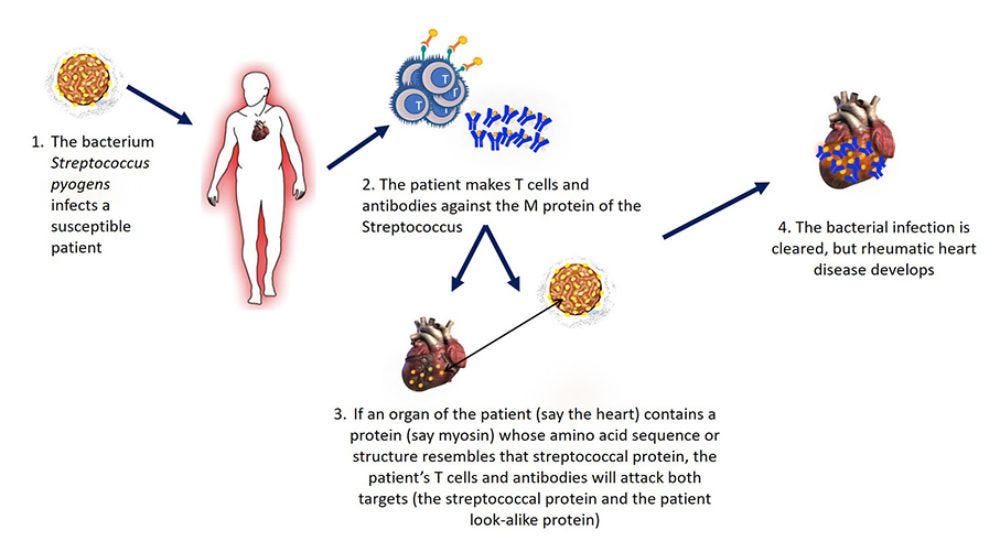
So you guys get it right? Self-directed cross-reactivity is not good.
I want to make an important distinction here regarding any conversation about SARS-CoV-2 spike, especially in the mRNA injection context. The thing that people are talking about with regard to injection-related spike embedded cell destruction, is not autoimmunity. It is the immune system working properly to clear cells with destroy-me flags on their surfaces. These ‘destroy-me’ flags are the (spike) peptides proudly displayed on cell surfaces by MHC molecules or can even be the spike embedded directly in the cell membrane. When a T or a B cell, for example, sees these flags, they do like Daleks: exterminate. EXTERMINATE! This is bad - by the way - for those cells. Every cell that gets embedded with the now known to be circulating spike is set for destruction by the immune system just doing its job. This is why it’s a bad thing when spike embeds epithelial cells lining blood vessels, cells that line capillaries in the brain and myocardial cells, to name a few places it’s been found to be embedded.
The idea behind autoimmunity development in the context of molecular mimicry is based on the development of these same T and B cells (and antibodies) as part of a normal immune response to foreign antigens. But, ‘by accident’, they can also potentially bind to and ‘act on’ epitopes that may not be the proteins of SARS-CoV-2 and, yes, that definitely includes the spike protein.
Autoimmunity itself involves loss of immunological tolerance that occurs when there is a failure of immune cells to recognize self antigens as ‘self’. This occurs somewhere during the process of tolerization in the thymus and the bone marrow that I describe here.
Autoimmunity as a whole describes an immunological attack on self proteins.
So to repeat - in the context of spike protein - the molecular mimicry aspect of autoimmunity relies on the well-established immunological concept that there are antigenic epitopes in the spike protein whereby through detection of spike as a foreign protein by the immune system, spike-specific T and B cells are produced en masse to fend off said foreign protein as part of an elaborate defense system. But accidentally, these T and B cells (and antibodies) also ‘establish’ the ability to bind other epitopes that kind of ‘look right’ (how you doin’?). These other epitopes might be found on your own proteins - as in, self proteins! Ouchie. What if those proteins were CNS proteins?
Back to the paper.
Scarily, the authors found a higher degree of peptide similarity between SARS-CoV-2 N and MS-associated proteins, than for 3 out of 4 seasonal coronaviruses that also had significant peptide similarities. This means, that yet again, something is ‘special’ about SARS-CoV-2 in this regard - myelin proteolipid protein (PLP) significantly overlapped only with the N protein of SARS-CoV-2: this might make the difference pathologically. Myelin proteolipid protein (PLP) is implicated in the development of MS, by the way.13141516

I had to shown this diagram from Figure 4 in the paper because I just love Venn Diagrams. Lots of other interesting proteins in there. They write:
We asked whether any of these proteins [driving immune responses in individuals recovering from SARS-CoV-2] shared significant homology with MS-associated neuro-antigens - an observation which would fortify the proposed case for molecular mimicry in the development of MS following SARS-CoV-2 infection. Interestingly, both the 9mer and 15mer groups (representing MHC I and II, respectively) from nucleocapsid showed significant sequence overlap with MS-associated proteins, in contrast to most of the other proteins tested in our analysis.
So indeed, there’s a case for molecular mimicry as the culprit here for development of MS via autoimmunity and it is SARS-nCoV-2-related. This only applies to the N protein, and somewhat to the replicase polyprotein1ab, for now.
I might write up “Lack of evidence of significant homology of SARS-CoV-2 spike sequences to myocarditis-associated antigens” tomorrow. It’s a paper that also came out recently with the guy who seems to be responsible for the PEPmatch bioinformatics tool, as first author. Seems like an interesting paper from quick scan.
Lake CM, Breen JJ. Sequence similarity between SARS-CoV-2 nucleocapsid and multiple sclerosis-associated proteins provides insight into viral neuropathogenesis following infection. Sci Rep. 2023 Jan 8;13(1):389. doi: 10.1038/s41598-022-27348-8. PMID: 36617594.
Multifunctional protein involved in the transcription and replication of viral RNAs. Contains the proteinases responsible for the cleavages of the polyprotein. https://pubchem.ncbi.nlm.nih.gov/protein/P0C6X7
Kuerten S, Lanz TV, Lingampalli N, Lahey LJ, Kleinschnitz C, Mäurer M, Schroeter M, Braune S, Ziemssen T, Ho PP, Robinson WH, Steinman L. Autoantibodies against central nervous system antigens in a subset of B cell-dominant multiple sclerosis patients. Proc Natl Acad Sci U S A. 2020 Sep 1;117(35):21512-21518. doi: 10.1073/pnas.2011249117. Epub 2020 Aug 18. PMID: 32817492; PMCID: PMC7474673.
Genain, C., Cannella, B., Hauser, S. et al. Identification of autoantibodies associated with myelin damage in multiple sclerosis. Nat Med 5, 170–175 (1999). https://doi.org/10.1038/5532.
Hedegaard CJ, Chen N, Sellebjerg F, Sørensen PS, Leslie RG, Bendtzen K, Nielsen CH. Autoantibodies to myelin basic protein (MBP) in healthy individuals and in patients with multiple sclerosis: a role in regulating cytokine responses to MBP. Immunology. 2009 Sep;128(1 Suppl):e451-61. doi: 10.1111/j.1365-2567.2008.02999.x. Epub 2008 Dec 18. PMID: 19191913; PMCID: PMC2753924.
Cusick MF, Libbey JE, Fujinami RS. Molecular mimicry as a mechanism of autoimmune disease. Clin Rev Allergy Immunol. 2012 Feb;42(1):102-11. doi: 10.1007/s12016-011-8294-7. PMID: 22095454; PMCID: PMC3266166.
Rojas M, Restrepo-Jiménez P, Monsalve DM, Pacheco Y, Acosta-Ampudia Y, Ramírez-Santana C, Leung PSC, Ansari AA, Gershwin ME, Anaya JM. Molecular mimicry and autoimmunity. J Autoimmun. 2018 Dec;95:100-123. doi: 10.1016/j.jaut.2018.10.012. Epub 2018 Oct 26. PMID: 30509385.
Segal Y, Shoenfeld Y. Vaccine-induced autoimmunity: the role of molecular mimicry and immune crossreaction. Cell Mol Immunol. 2018 Jun;15(6):586-594. doi: 10.1038/cmi.2017.151. Epub 2018 Mar 5. PMID: 29503439; PMCID: PMC6078966.
Number of people with MS | Atlas of MS. https://www.atlasofms.org/map/global/epidemiology/number-of-people-with-ms
Marrama, D., Mahita, J., Sette, A. & Peters, B. Lack of evidence of significant homology of SARS-CoV-2 spike sequences to myocarditis-associated antigens. eBioMedicine 75, (2022).
Sunil K. Joshi, Padma R. Suresh, Virander S. Chauhan; Flexibility in MHC and TCR Recognition: Degenerate Specificity at the T Cell Level in the Recognition of Promiscuous Th Epitopes Exhibiting No Primary Sequence Homology. J Immunol 1 June 2001; 166 (11): 6693–6703. https://doi.org/10.4049/jimmunol.166.11.6693
https://pathology.jhu.edu/autoimmune/causes/
Trotter, J. L. et al. T cell recognition of myelin proteolipid protein and myelin proteolipid protein peptides in the peripheral blood of multiple sclerosis and control subjects. J. Neuroimmunol. 84, 172–178 (1998).
Shaw, S. Y., Laursen, R. A. & Lees, M. B. Analogous amino acid sequences in myelin proteolipid and viral proteins. FEBS Lett. 207, 266–270 (1986).
Amor, S., Baker, D., Groome, N. & Turk, J. L. Identification of a major encephalitogenic epitope of proteolipid protein (residues 56–70) for the induction of experimental allergic encephalomyelitis in Biozzi AB/H and nonobese diabetic mice. J. Immunol. 150, 5666–5672 (1993).
Greer, J. Increased immunoreactivity to two overlapping peptides of myelin proteolipid protein in multiple sclerosis. Brain 120, 1447–1460 (1997).




Christina Applegate recently diagnosed with MS. Gotta wonder. People who were once healthy suddenly diagnosed with a disease. My thought has always been that these shots speed up anything that may be going on in one’s body already. It will be near impossible to blame these sudden onsets of diseases on the shots. They knew this when they created these bioweapons. They are even able to hide the obvious ones like we saw with Damar. My guess is that will say he had some cardio defect no one knew about that he was born with. It went undetected. Recently a young women died who had previously had her spleen removed due to a car accident and her sudden death became the result of a type of infection due to having no Spleen. They can make up anything for these sudden deaths. And they are!!
Thank you Jessica for another intriguing article.. it would be interesting to see a chart tracking the last 20 years increase in autoimmune disorders overlapped by one tracking the increase in vaccinations over same.. and a chart tracking the increase in pesticide, herbicide, fungicide use over 20 and one tracking the increase in all new Industrial chemicals over same.. and then last but not least the increase in EMFs over 20.. I bet the numbers would be daunting.. Our poor Immune system is under one hell of a siege.. Time to go as Organic as possible and back to Nature
Keep up your extraordinary work! You a Bad Rad Momma!