Is the spike protein acting as a prion with regard to hemoglobin molecules? And is porphyria being induced?
If the answers are 'yes', this is very bad news.
Strap in, hold onto your hats and grab a glass of wine. This is a long article. And it’s very important.
Coincidence?
Pfizer to Acquire Global Blood Therapeutics for $5.4 Billion to Enhance Presence in Rare Hematology
Monday, August 08, 2022 - 06:45am
On blood
Blood. Ahh blood. From silly B-rated horror films, to the first time we scraped our knees falling off of our bikes, we all know a little about blood. It runs. It clots. It’s red. It smells like… iron. Blood runs through our arteries and veins and is an essential component to our incredible living bodies and our circulatory systems. Blood is made up of white blood cells, platelets, plasma (that contains proteins, water, hormones, nutrients, etc) and red blood cells. The following delightful artistic depiction shows red blood cells (red), white blood cells (yellow) and activated platelets (green).

On red blood cells/erythrocytes
Red blood cells or erythrocytes, comprise a large component of the blood and originate from the bone marrow.1 Production of red blood cells is controlled by a kidney-sourced hormone called erythropoietin (meaning ‘make red’ from ancient Greek: ερυθρός/erythros(=red) + ποιέω/poieo (=create, make)). They come about from the bone marrow as immature cells, and take about 7 days to mature to end up circulating in the blood, where they ‘live’ about 120 days. They are anucleated (they do not contain a nucleus) so they can slip in and out of tight spaces like blood vessels within the circulatory system. Each cell contains millions of proteins called hemoglobin. Blood is red because of red blood cell content, and red blood cells are red because of the iron cores in the heme (porphyrin containing iron ring - I explain this soon) that make up the hemoglobin proteins.
“Specifically it is the iron (Fe) atom in the heme component that contributes with a bound Oxygen molecule to the red color.”2 Red blood cells also balance Nitric Oxide (NO) scavenging and production.3 Perhaps one of the most relevant characteristics of red blood cells is that they express Cluster of Differentiation molecule 147 (CD147) on their cell surfaces, which is a ligand of SARS-nCoV-2 spike protein.4 I will write more on this in the following sections.
Red blood cells also carry blood group antigens on their surfaces. This means that depending on which blood type you are, you will have specific antigens on the surfaces of your red blood cells.
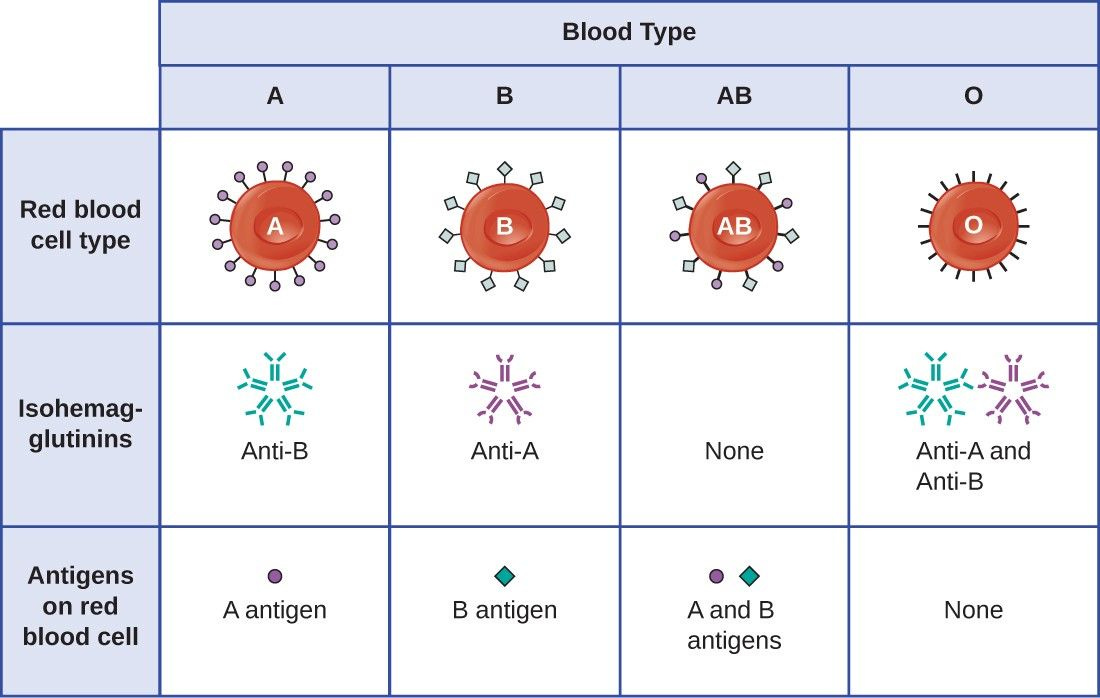
If you are blood type O, then you do not have blood group antigens on your red blood cells, but you do have anti-A and B isohemagglutinins (antibodies) against A and B antigens. That’s why you can’t get a blood transfusion from someone who doesn’t have blood type O, if you are blood type O, for example. Your ‘O’ anti-A and B isohemagglutinins will recognize the foreign blood group antigens on those red blood cells and destroy them (hemolysis). Blood type O (Rh D-) individuals are the so-called ‘universal donors’, therefore: they can give to anyone because they lack antigens on their red blood cell surfaces. Blood type ABers (Rh D+), the so-called universal recipients, can get blood from everyone, because they don’t have attack antibodies! Very cool, eh? *One must also consider the positivity or negativity of the Rhesus (Rh) D antigen, but never mind this for now.
I wrote a little on blood group antigens in this article because there have been studies that have shown that the seriousness of COVID-19 pathology can be associated with blood type.5 6
Many studies report that blood type A might predispose one to increased susceptibility of infection with SARS-CoV-2, and type O and Rh-negative blood groups might be protective.
On hemoglobin
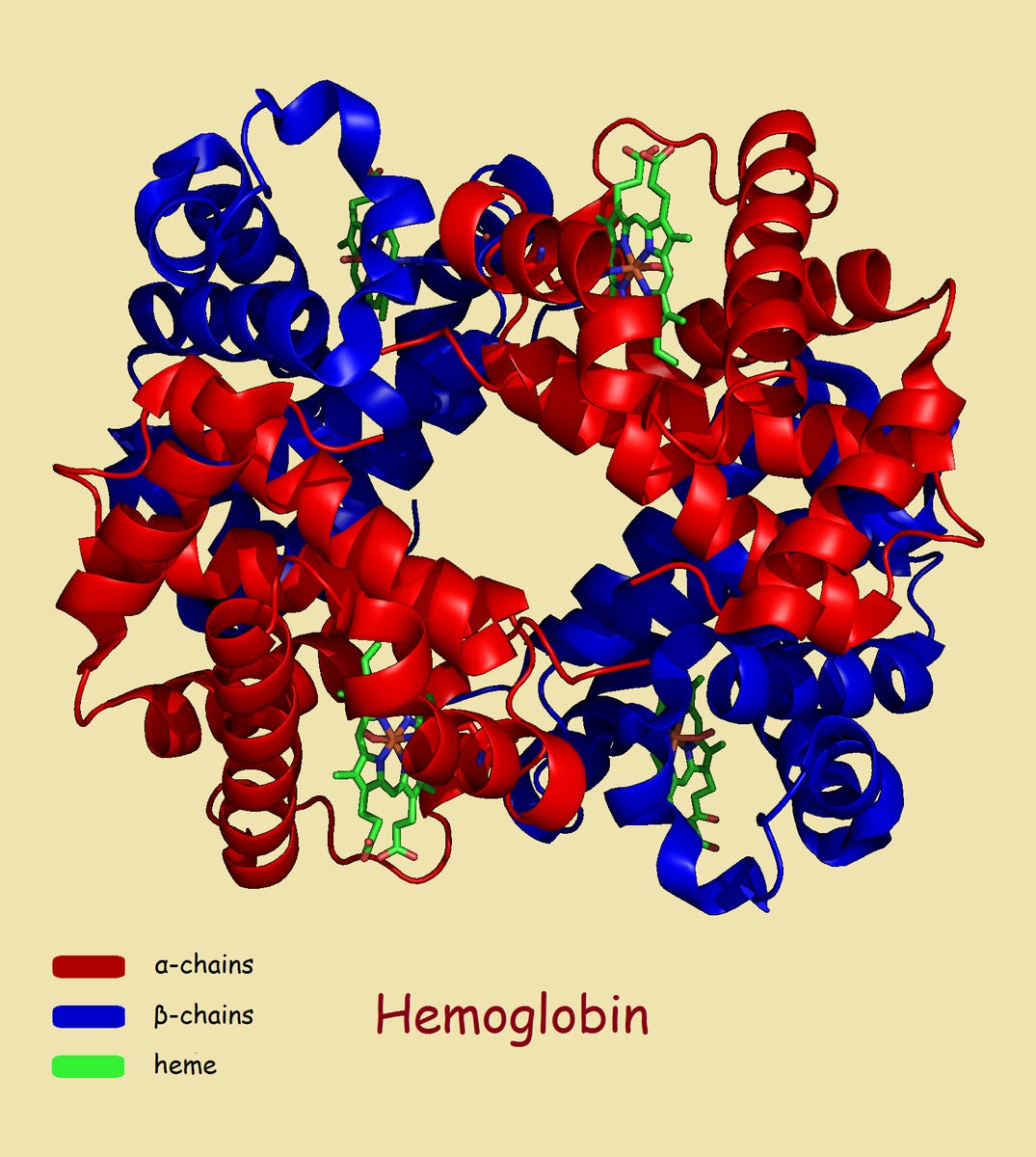
Hemoglobin is made up of heme and globin. Go figure. Heme is a porphyrin ring with an iron at its core and globin is a lump - from the Sanskrit: ग्लुन्थ (gluntha, “lump”). Hemoglobin is a protein found in red blood cells or erythrocytes (~270 million per cell) that binds oxygen molecules. Each hemoglobin protein is composed of four globin subunits: 2 alpha and 2 beta, and each subunit has an iron-bound heme (a porphyrin ring), whereby each subunit is capable of binding/capturing oxygen atoms - depending on the state of the iron.
As oxygens bind or are captured (however you want to perceive it), the hemoglobin protein undergoes conformational shifts, as per subunit, that increases the capture-ability of additional oxygen molecules in each subsequent subunit. The hemoglobin’s job is to deliver oxygen to sites in the human body, like capillaries, for cellular metabolism. Human cells cannot produce Adenosine Triphosphate (ATP) energy molecules in the mitochondria of the cells for use in the vital functions of the body without oxygen. Oxygen release from the heme complex is dependent on the pH of the blood. When carbon dioxide levels are high in the blood, the pH of the blood drops (it becomes more acidic because there are more hydrogen ions) and this causes a conformational change in the hemoglobin protein promoting the release of the oxygens from the heme complexes. The oxygens diffuse into muscle cells, get transported to mitochondria and ATP is produced. It’s kind of like tickling your sister to make her let go of the apple you want to eat. You’re the pH, she’s the hemoglobin, her hand is the heme and the apple is the oxygen.
It is important to know for later on in this article, that pathogens can cause hemoglobin to ‘denature’ or unravel. Imagine a denatured protein like a string of beads bunched up in a shape in your hand being straightened out by simply holding the string from end to end. This denaturing of hemoglobin causes the release of the porphyrin ring and the iron causing ‘disease’ states. This is one of the things that makes the malarial parasite Plasmodium falciparum so devastating to humans: it eats hemoglobin for lunch. And dinner.7 Bacterial pathogens intentionally induce hemolysis to get the iron for themselves.8 Other invaders do this as well. This is like when government ‘officials’ want your heart because they don’t have one so they take yours and then you explode. Wait, that’s not a good example. Is it?
Eukaryotic pathogens, including Leishmania, Entamoeba, and Trypanosoma, have evolved convergent mechanisms of heme-iron acquisition from this abundant host molecule. Protozoa capture hemoglobin through either specific surface receptors or phagocytosis. (Reference #8)
It has also been suggested that viral pathogens can cause dissociation of hemoglobin. SARS-nCoV-2 is one of these suggested viral pathogens. It was found in a recent study that ORF3a, E and N of SARS-nCoV-2 have heme linked sites.9 The authors also provide lists of agents that bind these sites to prevent the potential stealth stealing of heme from the porphyrin ring. Read the above article. It is insane. In a good way.
On porphyrins
Porphyrins are the conjugate acids of ligands that bind metals to form complexes like heme.10 Heme is a porphyrin with an iron (Fe) at its center. In Figure 5, it is an Fe2+ indicative of the fact that it can bind Oxygen.11 The hemoglobin protein, of course, is made up of heme and globin.
Porphyrin is constructed via a complex mutli-stage process (the porphyrin development pathway) that starts with 8 molecules of Aminolevulinic acid (ALA), Succinyl-CoA and glycine and ends with porphyrin. I am constantly amazed that anything works in biology. There are so many ways that things can go wrong. Which, is again, why I don’t understand why there are so many exceedingly arrogant humans who think it’s a good idea to mess with biology.
On porphyria
Porphyria is an accumulation of porphyrins in the liver or bone marrow due to defects in the porphyrin development pathway. Porphyrias are rare diseases. Studies suggest that all types of porphyrias combined affect fewer than 200,000 people in the United States, and that these most common form occurs at a rate of 1/10,000, and the least common at a rate of 1/1,000,000.12 13 14 15 Acute porphyria is more common in females than in males and often begins when people are between the ages of 15 and 45.16 Sounds like the millennial age group, doesn’t it? Just saying. Let’s hold that thought for now.
On prions
Please go back and read this article to get background on prions.
These mis-folded proteins are called prions (mis-folded prion protein (PrPSc - Sc is for Scrapie2 3)) and are associated with neuro-degenerative diseases like Alzheimer’s disease. It seems that they can transform or ‘teach’ other prion proteins to mis-fold (transmissible), as well. It’s not really teaching as much as proximal disallowance of proper folding. This ‘enables’ an autocatalytic reaction whereby the PrPSc catalyzes the mis-folding of PrPc to produce more PrPSc due to the fact that prions are resistant to proteolysis - the process of ‘removal’ of mis-folded proteins.
Prions can induce other proteins to become mis-folded.
On prion-like spike protein
Please go here and here and here and especially here for my previously written articles for background related to the prion-like spike protein. It’s well documented in the literature as a potential serious problem. For references, head to my Substack articles.
Alright, now that we have all that covered, let’s get clinical.
On Malaria and COVID-19
COVID-19 and malaria (these names refer to the diseases caused by the virus SARS-nCoV-2 and the unicellular protozoan obligate parasite Plasmodium falciparum that manifest clinically, respectively) are more alike than any of us may think.17 18 They are very, very different. One is a complex ‘virus’ and one is a single-celled creature. They don’t look the same at all! How can these guys be alike? Well, they can be alike in the physiological effects that they impose on biological entities known as humans.

Recent evidence shows that SARS-CoV-2 is capable of affecting the genetics and dynamics of erythrocytes and this coexists with a non-homeostatic function of cardiovascular, respiratory and renal systems during COVID-19. In hypothesis, SARS-CoV-2-induced systematical alterations of erythrocytes dynamics would constitute a set point for COVID-19-related multiple organ failure syndrome and death. (Reference #17)
Here are 4 facts:
Plasmodium falciparum is the etiological agent of malaria in humans and does its damage by infecting red blood cells thus causing extensive changes in them and can ultimately result in hemolysis.19
Plasmodium falciparum can enter red blood cells via CD147.20 21 (Throat clearing: SARS-nCoV-2 infects cells via CD147 as well - References #31 and #32)
Once inside red blood cells, Plasmodium falciparum feasts on hemoglobin. (Ahem: SARS-nCoV-2 disrupts hemoglobin levels22 (Reference #9 shows inhibition of heme metabolism by SARS-nCoV-2.))
Hemolysis causes a reduction in oxygen supply to all the places in the body that need oxygen - which is literally, all of the places: no oxygen supply = death.
Malaria is curable using (Hydroxy)chloroquine.23 24 25 (Ahem: Yeah. You know.26 27)
“Antimalarial drugs such as chloroquine and possibly artemisinin inhibit hemoglobin detoxification by Plasmodium, underscoring the importance of this process for malarial viability”.28 29 And absolutely amazingly, “the susceptibility of individuals to bacterial infections (like from Staphylococcus aureus) may be affected by hemoglobin polymorphisms.”30 This is just a point of interest to me on hemoglobin polymorphisms.
Alright, you saw my saucy ‘throat-clearing’ points. So let’s ask some questions. We’re allowed to do that here.
Question: Since Plasmodium falciparum can use CD147 to enter erythrocytes to disassemble hemoglobin to induce a disease state (and thus induce amyloid plaque production (reference #40)), and since SARS-nCoV-2 can also use CD147 to enter erythrocytes31 32, does it also ‘disrupt’ hemoglobin (or even to ‘attack the beta chains of hemoglobin’ (reference #9)) to induce disease and potentially amyloid plaques?33 34
If you’ve been paying attention, you might be thinking right about now “do individuals with malaria suffer from amyloid plaque formation?”. And you would be thinking clearly! It has been shown that amyloidogenic peptides encoded in the Plasmodium organism play a role in malaria pathology.35 So it makes even more sense to me that since we already know that the spike protein contains amyloidogenic peptides, that the pathology of COVID-19 would involve amyloid plaque formation based both on the amount of protein produced/present and the amount of damage to the red blood cells.
Let’s ask some bigger questions now.
Question #1: Can the prion-like protein associated with the SARS-nCoV-2 spike protein induce mis-folding of the hemoglobin molecule to incapacitate oxygen binding either in the context of SARS-nCoV-2 or the injection-produced spike?
Question #2: Does the spike protein induce denaturation of the hemoglobin protein by reducing it to its apohemoglobin form (no heme) and thus allowing it to become amyloidogenic?36 37
Before we continue with trying to answer these 2 questions, let’s go back to malaria for a second and ask another question.
Question: What effect does having had malaria previously have on SARS-nCoV-2 introduction? And what about co-infection with malaria and SARS-nCoV-2?
Just as a point of absolute hilarity,
The recovery of SARS-CoV-2 infection in HCWs was faster (mean 8 days) with co-infection of malaria than without malaria (p < .005).38
So you recover faster from SARS-nCoV-2 if you have malaria. It has been shown that the reason why people in malarial endemic regions do better with COVID-19, is due to cross-reactive T cells.39 This is incredibly important and relevant but I am going to focus from here on out on trying to answer questions #1 and #2. I just needed to put that in there because of the obvious connection and the chloroquine thing. I will return to that.
Let’s get back to whether or not SARS-nCoV-2 buggars hemoglobin. It has been shown that SARS-nCoV-2 attacks the 1-Beta Chain of Hemoglobin and Captures the Porphyrin to Inhibit Heme Metabolism in this paper. (Reference #9)
Amyloid fibrils have been shown to develop from hemolysis of hemoglobin.40 As a reminder, amyloid fibrils are insoluble protein nanofibers that spontaneously accumulate, or self-assemble, to form amyloid plaques and ‘disease’. The authors of this paper concluded that:
This study conclusively demonstrates the formation of amyloid fibrils from hemoglobin for the first time, and also introduces a cost-effective method for amyloid fibril manufacture using meat industry by-products.
This frightens me: this particular application to biotech and the potential for harms in vivo. Despite the fact that the authors claim that these structures have amazing applications in real life as versatile and durable nano-materials, I still remain concerned. I would want to know the answers to some basic questions before further studies were conducted. What would happen, for example, if a ‘substance’ was injected into humans that could denature hemoglobin? Would this result in the aggregation of fibrils and disseminated amyloid plaques? Are there conditions or disease states in humans that equate to this? Well, we know that Plasmodium falciparum equates to this. And we also have strong evidence here that SARS-nCoV-2 equates to this. And what of Alzheimer’s and Parkinson’s?
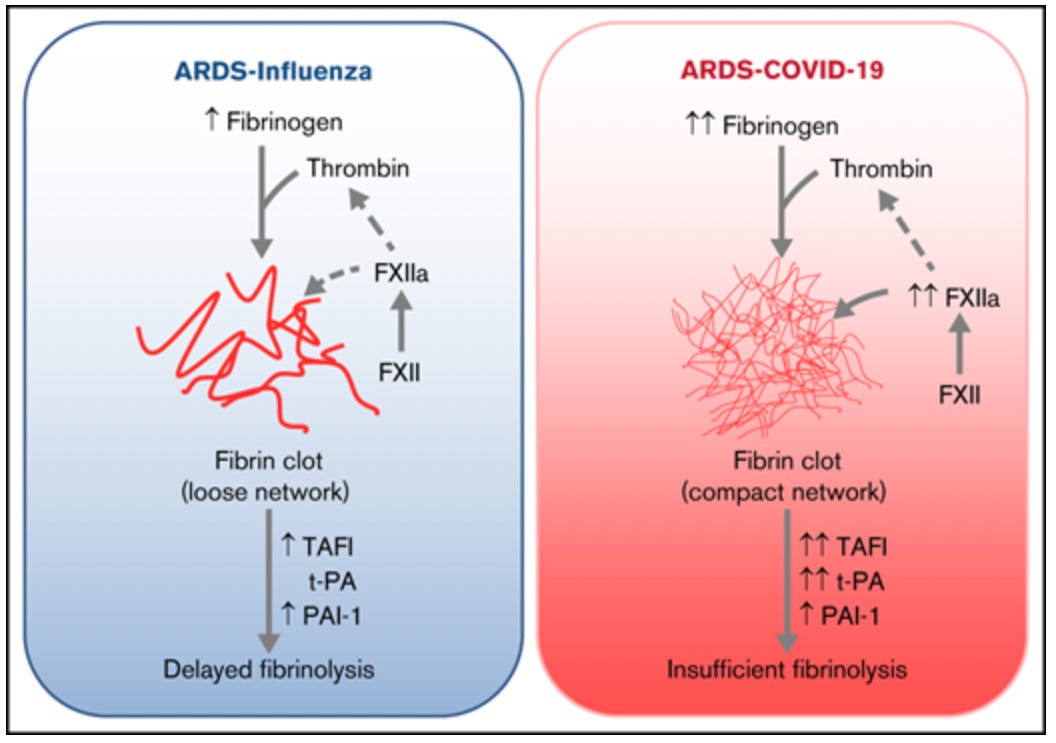
Scarily, it has been shown in COVID-19 patients that the fibrinolytic system is dysregulated, “as evidenced by elevated levels of thrombin-activatable fibrinolysis inhibitor, tissue-plasminogen activator, and plasminogen activator inhibitor-1 in COVID-19 potentiated this effect.”41 Fibrin clots were compared in individuals with ARDS associated with influenza versus ARDS associated with COVID-19, and it was shown that the clots in the COVID-19 cases were much denser and the reason for this came down to a reduced ability to break up clots (fibrinolysis).
Think of it this way. You have a bunch of life-savers - the candy, not the life-guard apparatus. You stick each one in your mouth, give them each a go, decide you don’t like the taste of any of them, and so you stick them together making a gross, sticky life-saver clump. Then, for some reason, you wrap the clump in string. Then your mom comes home and sees this and freaks out and tells you to clean it up. To clean up this mess, you have to unwrap the string or find a way to dissolve it. But, lo and behold, you can’t do either of these things, for some reason. Maybe you don’t have any acid left to dissolve the string. And maybe you shouldn’t be playing with acid. So, what happens if you can’t clean up your mess?
Media break: I recommend watching this video to get a better idea of the normal clotting/unclotting pathway in humans. This guy won’t have 2 subscribers for much longer. Everyone needs to know why it’s essential not to introduce potential agents to disrupt this pathway. By the way, the so-called ‘coagulation cascade’ is linked to heparin. I wrote about this as well here.
On clinical reports to help answer questions #1 and #2
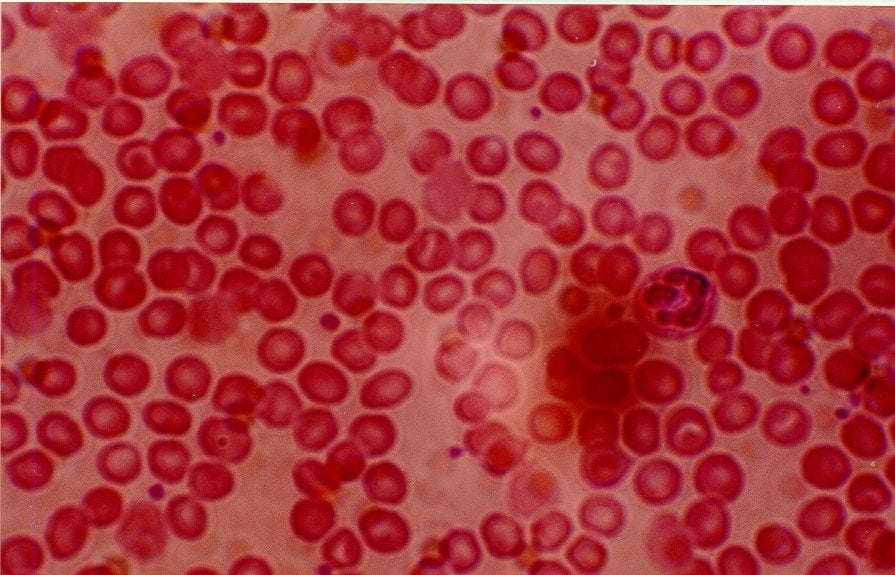
A logical way that I can think of to provide some clinical evidence to help answer questions 1 and 2, is to look for something called Heinz bodies in red blood cells composed of denatured hemoglobin.42 Heinz bodies, named after Robert Heinz in the late 1800s, are round inclusions that are formed by damage to the hemoglobin component molecules, usually through oxidative damage by administered drugs, or can be from an inherited mutation.43
Dysregulation in red blood cell function in the context of SARS-nCoV-2 has been shown in recent publications where Heinz bodies were discovered in 14 individuals with severe COVID-19.44 45 They look like the following in Figure 9.

This means that the hemoglobin component in the red blood cells of these individuals was denatured. So even though we can visibly see the effect of the virus on hemoglobin in red blood cells, what I would like to see now is evidence that shows Heinz bodies following COVID-19 injection to provide evidence of spike-induced red blood cell damage. I couldn’t find anything relating to Heinz bodies specifically in the literature but I did find evidence of hemolysis.46 The authors of this work suggest that the post injection hemolysis was mediated by complement activation and not the direct effect of the spike protein. They could be right.
So I am coming to this end of this amazing introductory journey through red blood cell characterization in the context of pathogens. I want to leave the questions ‘hanging’ to allow us all to think and because I am sure the complete answers will come soon. I also want to end with VAERS evidence. Why not. It’s what I do.
There is no MedDRA code for ‘Heinz body’ so I decided to check for other ‘hemoglobin issues’ and oxygen saturation issues including ‘Haemoglobin decreased’ and ‘Oxygen saturation decreased’ to look for reported clinical associations with hemolysis. Here’s what the query of the Domestic and Foreign data returned.

The absolute count of reports is N = 16,484 and when normalized to CDC single dose data, we get Figure 10. The data indicate that it is primarily our elders affected, but not exclusively: no one is immune to the effects. And remember, our elders have been being injected for the longest timeframe.
The leaves rustling in the wind is indicative of the hemolysis wind, but we need more studies to determine whether or not there is a propensity toward Heinz bodies in COVID-19 injected people. Anybody want to launch a trial? If we can determine that there is a propensity, then we have evidence to support the theory that the prion-like spike protein may be causing hemoglobin proteins to mis-fold.
Let’s answer one more question since I introduced you all to porphyrins.
#3. Does the spike protein from the COVID-19 injections cause porphyria?
By the way, for more advanced and awesome reading on hemoglobin stability, you can read this paper published in 1970.47 The oldies are often the goodies.
I would say, yes, the COVID-19 injections are causing porphyria. They certainly are associated with increased reporting (a 17,265% increase as compared with the average of the past 5 years) (Figure 12) and the reports are filed in temporal proximity to the injection dates (Figure 13). A VAERS query truly shocked me. Look at the reporting rate without the underreporting factor! N = 23,367! So yeah, it appears as though the spike protein may be inducing porphyria.
Not comparable to the past 5 years.
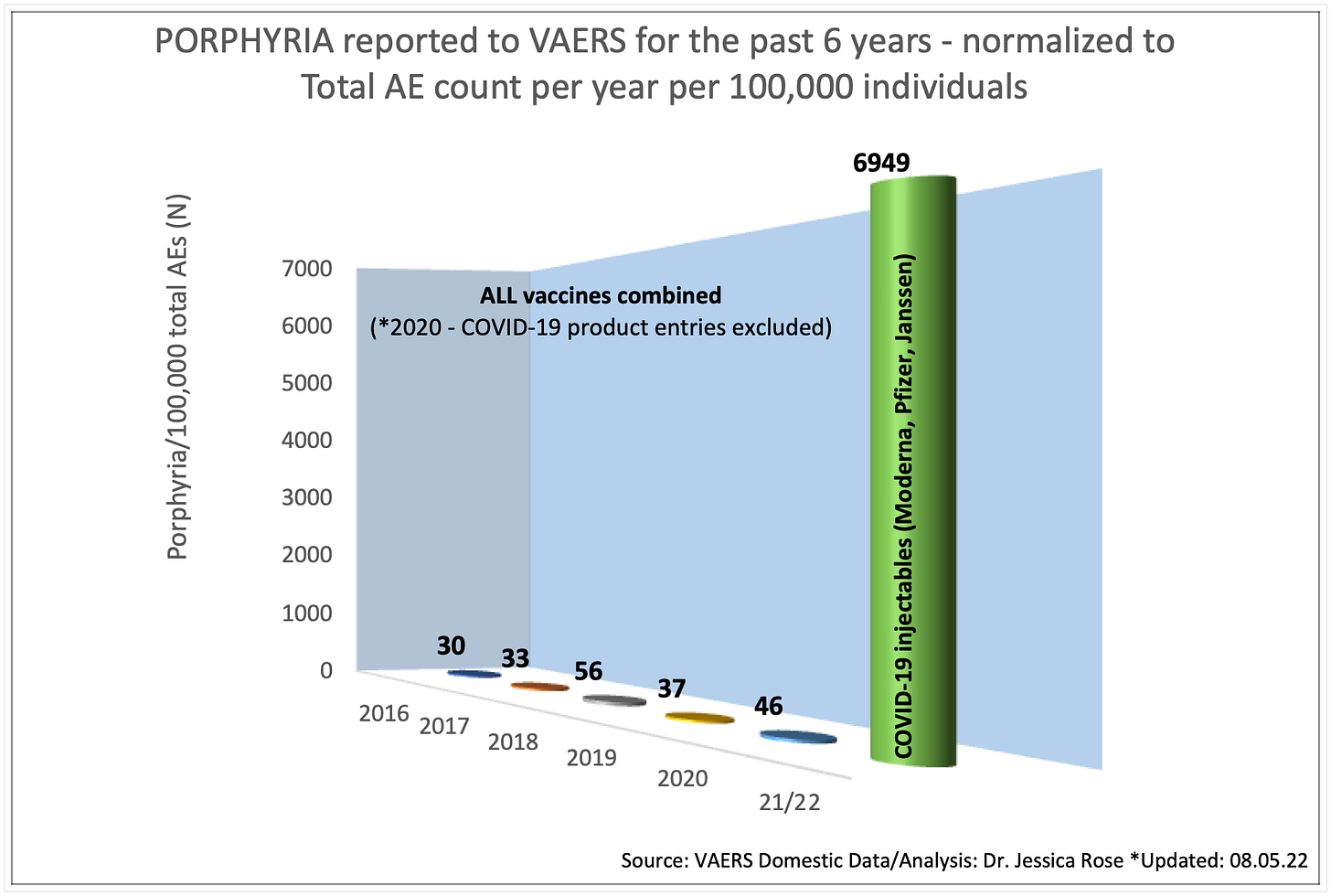
And the reports are highly temporally-associated with injection date.

So the bottom line of all of this information is this: the virus infects the RBCs using spike protein via the CD147 receptor on red blood cells which causes hemolysis (rupture of the red blood cell). This causes the release of massive amounts of hemoglobin. Then the spike protein, due to its amyloidogenic peptides, triggers mis-folding of the hemoglobin into amyloid fibrils causing subsequent blood clots. The blood clots would be enhanced due to antibodies (Ag:Ab complexes). So a big remaining question that needs to be answered is: Under what circumstances can and does hemolysis occur in the context of the COVID-19 injections due to the spike protein?
On Solutions
Demand full blood counts and use this to predict outcome.48 Set up Heinz body study. Use hydroxychloroquine if exposed to SARS-nCoV-2. STOP INJECTING YOURSELF. At least until we have more answers to these very concerning questions.
Open-ended questions:
Question #1: What happens to individuals with hemochromatosis when exposed to SARS-nCoV-2?
Question #2: Is hydroxychloroquine more effective in the Delta context and less so in the Omicron context due to differences in pathologies associated with hemoglobin dissociation linked to the spike sequence?
Question #3: Is the systemic micro-clotting we are seeing clinically and in pharmacovigilance databases like VAERS, in fact, due to hemolysis from the effects of the spike protein?
Biggie question master question: Can the prion-like protein associated with the SARS-nCoV-2 spike protein induce mis-folding of the hemoglobin molecule to incapacitate oxygen binding either in the context of SARS-nCoV-2 or the injection-produced spike?
I think the answer is ‘yes’. And I think this is precisely why oxygen stats decline, why doctors were comparing the clinical signs of Delta to altitude sickness, why vents actually killed more people than they ever could have saved (they were not used properly), why porphyria rates are through the roof, why micro-clotting is notoriously-associated with COVID-19, and why some people die.
The soft, spongy tissue that has many blood vessels and is found in the center of most bones. There are two types of bone marrow: red and yellow. Red bone marrow contains blood stem cells that can become red blood cells, white blood cells, or platelets. Yellow bone marrow is made mostly of fat and contains stem cells that can become cartilage, fat, or bone cells. https://www.cancer.gov/publications/dictionaries/cancer-terms/def/bone-marrow
https://www.quora.com/profile/Carl-Malmgren
C. Helms et al. Erythrocytes and Vascular Function: Oxygen and Nitric Oxide. Front. Physiol., 22 February 2018. Sec. Vascular Physiology. https://www.frontiersin.org/articles/10.3389/fphys.2018.00125/full
Wang, Ke & Chen, Wei & Zhou, Yu-Sen & Lian, Jian-Qi & Zhang, Zheng & Du, Peng & Gong, Li & Zhang, Yang & Cui, Hongyong & Geng, Jie-Jie & Wang, Bin & Sun, Xiu-Xuan & Wang, Chun-Fu & Yang, Xu & Lin, Peng & Deng, Yong-Qiang & Wei, Ding & Yang, Xiang-Min & Zhu, Yu-Meng. (2020). SARS-CoV-2 invades host cells via a novel route: CD147-spike protein. 10.1101/2020.03.14.988345.
Kim Y, Latz CA, DeCarlo CS, Lee S, Png CYM, Kibrik P, Sung E, Alabi O, Dua A. Relationship between blood type and outcomes following COVID-19 infection. Semin Vasc Surg. 2021 Sep;34(3):125-131. doi: 10.1053/j.semvascsurg.2021.05.005. Epub 2021 Jul 18. PMID: 34642032; PMCID: PMC8286549.
Al-Youha SA, Alduaij W, Al-Serri A, et al. The impact of ABO blood groups on clinical outcomes and susceptibility to COVID-19: A retrospective study in an unselected population. Transfusion. 2021;61(5):1631-1641. doi:10.1111/trf.16365.
Goldberg DE, Slater AF, Cerami A, Henderson GB. Hemoglobin degradation in the malaria parasite Plasmodium falciparum: an ordered process in a unique organelle. Proc Natl Acad Sci U S A. 1990 Apr;87(8):2931-5. doi: 10.1073/pnas.87.8.2931. PMID: 2183218; PMCID: PMC53807.
Pishchany, G., & Skaar, E. P. (2012). Taste for Blood: Hemoglobin as a Nutrient Source for Pathogens. PLoS Pathogens, 8(3), e1002535. doi:10.1371/journal.ppat.1002535 10.1371/journal.ppat.1002535.
Liu and Li. COVID-19: Attacks the 1-Beta Chain of Hemoglobin and Captures the Porphyrin to Inhibit Heme Metabolism. https://chemrxiv.org/engage/api-gateway/chemrxiv/assets/orp/resource/item/60c74fa50f50db305139743d/original/covid-19-attacks-the-1-beta-chain-of-hemoglobin-and-captures-the-porphyrin-to-inhibit-human-heme-metabolism.pdf
https://en.wikipedia.org/wiki/Porphyrin
https://chem.libretexts.org/Bookshelves/General_Chemistry/Book:_Structure_and_Reactivity_in_Organic_Biological_and_Inorganic_Chemistry_(Schaller)/V:__Reactivity_in_Organic_Biological_and_Inorganic_Chemistry_3/04:_Oxygen_Binding_and_Reduction/4.02:_Oxygen_Binding
https://www.niddk.nih.gov/health-information/liver-disease/porphyria
Ramanujam VS, Anderson KE. Porphyria diagnostics—part 1: a brief overview of the porphyrias. Current Protocols in Human Genetics. 2015;86:17.20.1–17.20.26. doi: 10.1002/0471142905.hg1720s86.
Bissell DM, Anderson KE, Bonkovsky HL. Porphyria. New England Journal of Medicine. 2017;377(9):862–872. doi: 10.1056/NEJMra1608634.
Balwani M, Bloomer J, Desnick R; Porphyrias Consortium of the NIH-Sponsored Rare Diseases Clinical Research Network. Erythropoietic protoporphyria, autosomal recessive. In: Adam MP, Ardinger HH, Pagon RA, et al, eds. GeneReviews [Internet]. University of Washington, Seattle; 1993–2020. Updated September 7, 2017. Accessed August 6, 2020.
Bissell DM, Wang B. Acute hepatic porphyria. Journal of Clinical and Translational Hepatology. 2015;3(1):17–26. doi: 10.14218/JCTH.2014.00039.
Mendonça MM, da Cruz KR, Pinheiro DDS, et al. Dysregulation in erythrocyte dynamics caused by SARS-CoV-2 infection: possible role in shuffling the homeostatic puzzle during COVID-19. Hematol Transfus Cell Ther. 2022;44(2):235-245. doi:10.1016/j.htct.2022.01.005.
Hussein MIH, Albashir AAD, Elawad OAMA, Homeida A. Malaria and COVID-19: unmasking their ties. Malar J. 2020;19(1):457. Published 2020 Dec 23. doi:10.1186/s12936-020-03541-w.
Mohandas N, An X. Malaria and human red blood cells. Med Microbiol Immunol. 2012 Nov;201(4):593-8. doi: 10.1007/s00430-012-0272-z. Epub 2012 Sep 11. PMID: 22965173; PMCID: PMC3699179.
Pretini Virginia, Koenen Mischa H., Kaestner Lars, Fens Marcel H. A. M., Schiffelers Raymond M., Bartels Marije, Van Wijk Richard. Red Blood Cells: Chasing Interactions. Front. Physiol., 31 July 2019. Sec. Red Blood Cell Physiology. https://doi.org/10.3389/fphys.2019.00945
Crosnier, C. et al. Basigin is a receptor essential for erythrocyte invasion by Plasmodium falciparum. Nature 480, 534-537 (2011).
Lippi G, Mattiuzzi C. Hemoglobin value may be decreased in patients with severe coronavirus disease 2019. Hematol Transfus Cell Ther. 2020 Apr-Jun;42(2):116-117. doi: 10.1016/j.htct.2020.03.001. Epub 2020 Apr 2. PMID: 32284281; PMCID: PMC7128154.
Coy D. Fitch and Natrice V. Russell. Accelerated Denaturation of Hemoglobin and the Antimalarial Action of Chloroquine. ASM Journal. Antimicrobial Agents and Chemotherapy. Vol. 50, No. 7.
White NJ. The treatment of malaria. N Engl J Med. 1996;335(11):800-806. doi:10.1056/NEJM199609123351107.
Slater AF. Chloroquine: mechanism of drug action and resistance in Plasmodium falciparum. Pharmacol Ther. 1993;57(2-3):203-235. doi:10.1016/0163-7258(93)90056-j.
de Reus YA, Hagedoorn P, Sturkenboom MGG, et al. Tolerability and pharmacokinetic evaluation of inhaled dry powder hydroxychloroquine in healthy volunteers. PLoS One. 2022;17(8):e0272034. Published 2022 Aug 5. doi:10.1371/journal.pone.0272034.
https://covid19criticalcare.com/covid-19-protocols/i-recover-long-covid-treatment/
Ziegler J, Linck R, Wright DW (2001) Heme Aggregation inhibitors: antimalarial drugs targeting an essential biomineralization process. Curr Med Chem 8: 171–189. 20.
Klonis N, Crespo-Ortiz MP, Bottova I, Abu-Bakar N, Kenny S, et al. (2011) Artemisinin activity against Plasmodium falciparum requires hemoglobin uptake and digestion. Proc Natl Acad Sci U S A 108: 11405–11410.
Pishchany G, McCoy AL, Torres VJ, Krause JC, Crowe JE, Jr., et al. (2010) Specificity for human hemoglobin enhances Staphylococcus aureus infection. Cell Host Microbe 8: 544–550.
Wang, K., Chen, W., Zhang, Z., Deng, Y., Lian, J. Q., Du, P., Wei, D., Zhang, Y., Sun, X. X., Gong, L., Yang, X., He, L., Zhang, L., Yang, Z., Geng, J. J., Chen, R., Zhang, H., Wang, B., Zhu, Y. M., Nan, G., … Chen, Z. N. (2020). CD147-spike protein is a novel route for SARS-CoV-2 infection to host cells. Signal transduction and targeted therapy, 5(1), 283. https://doi.org/10.1038/s41392-020-00426-x.
Ulrich H, Pillat MM. CD147 as a Target for COVID-19 Treatment: Suggested Effects of Azithromycin and Stem Cell Engagement. Stem Cell Rev Rep. 2020 Jun;16(3):434-440. doi: 10.1007/s12015-020-09976-7. PMID: 32307653; PMCID: PMC7167302.
https://news.cuanschutz.edu/news-stories/attack-on-red-blood-cells-a-prime-suspect-in-covids-debilitating-effects
Mendonça MM, da Cruz KR, Dos Santos Silva FC, Fontes MAP, Xavier CH. Are hemoglobin-derived peptides involved in the neuropsychiatric symptoms caused by SARS-CoV-2 infection? [published online ahead of print, 2022 Jul 27]. Braz J Psychiatry. 2022;10.47626/1516-4446-2021-2339. doi:10.47626/1516-4446-2021-2339.
Moles E, Valle-Delgado JJ, Urbán P, et al. Possible roles of amyloids in malaria pathophysiology. Future Sci OA. 2015;1(2):FSO43. Published 2015 Sep 1. doi:10.4155/fso.15.43.
This is the form without heme - porphyrin ring or iron. https://biochemden.com/heme-synthesis/
S. A. Kim, V. O. M. Villa & J. R. Hess (1994) Denatured Hemoglobin Increases Human Blood Mononuclear Cell Procoagulant Effect, Artificial Cells, Blood Substitutes, and Biotechnology, 22:3, 625-631, DOI: 10.3109/10731199409117892.
Mahajan NN, Gajbhiye RK, Bahirat S, et al. Co-infection of malaria and early clearance of SARS-CoV-2 in healthcare workers. J Med Virol. 2021;93(4):2431-2438. doi:10.1002/jmv.26760.
Iesa MAM, Osman MEM, Hassan MA, et al. SARS-CoV-2 and Plasmodium falciparum common immunodominant regions may explain low COVID-19 incidence in the malaria-endemic belt. New Microbes New Infect. 2020;38:100817. doi:10.1016/j.nmni.2020.100817.
Jayawardena N, Kaur M, Nair S, Malmstrom J, Goldstone D, Negron L, Gerrard JA, Domigan LJ. Amyloid Fibrils from Hemoglobin. Biomolecules. 2017 Apr 11;7(2):37. doi: 10.3390/biom7020037. PMID: 28398221; PMCID: PMC5485726.
Wygrecka M.et al; Altered fibrin clot structure and dysregulated fibrinolysis contribute to thrombosis risk in severe COVID-19. Blood Adv 2022; 6 (3): 1074–1087. doi: https://doi.org/10.1182/bloodadvances.2021004816
https://www.sciencedirect.com/topics/medicine-and-dentistry/heinz-body
https://en.wikipedia.org/wiki/Heinz_body
Mendonça MM, da Cruz KR, Pinheiro DDS, et al. Dysregulation in erythrocyte dynamics caused by SARS-CoV-2 infection: possible role in shuffling the homeostatic puzzle during COVID-19. Hematol Transfus Cell Ther. 2022;44(2):235-245. doi:10.1016/j.htct.2022.01.005.
Perrin and Gérard. Heinz bodies in COVID-19. https://onlinelibrary.wiley.com/doi/epdf/10.1111/ijlh.13926
Gloria F. Gerber, Xuan Yuan, Jia Yu, Benjamin A. Y. Cher, Evan M. Braunstein, Shruti Chaturvedi, Robert A. Brodsky; COVID-19 vaccines induce severe hemolysis in paroxysmal nocturnal hemoglobinuria. Blood 2021; 137 (26): 3670–3673. doi: https://doi.org/10.1182/blood.2021011548.
Rieder RF. Hemoglobin stability: observations on the denaturation of normal and abnormal hemoglobins by oxidant dyes, heat, and alkali. J Clin Invest. 1970;49(12):2369-2376. doi:10.1172/JCI106456.
https://www.nationalheraldindia.com/health/full-blood-count-can-predict-disease-severity-of-covid-patients


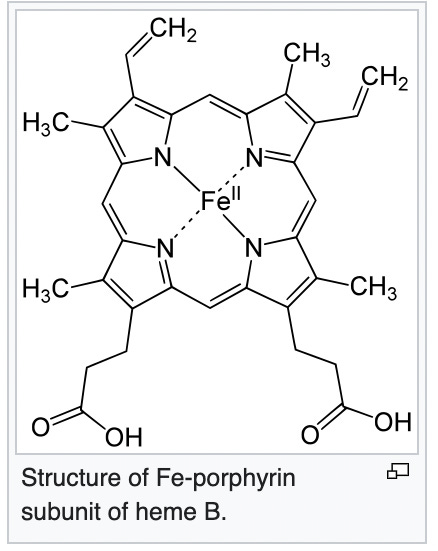

" I am constantly amazed that anything works in biology. There are so many ways that things can go wrong. Which, is again, why I don’t understand why there are so many exceedingly arrogant humans who think it’s a good idea to mess with biology. "
1. Excellent comment
2. It is all my fault; 100% hubris...
I'm convinced. You should publish this is a clinical journal.