I wrote a little Twitter teaser the other day about the wonderfulness of the newest smallpox/monekypox vaccine. Here, I elaborate with some fun facts in the latest, greatest FDA document.




But first, what is JYNNEOS, you ask? Well, you’ll know soon enough! It’s new! (no it’s not) It’s going to save you from everything! (no it won’t) It’s safe! (safety data questionable - funny that - especially for people with health concerns) It’s not a smallpox vaccine in the experimental phase! (yes it is)
Please go the BLA Clinical Review Memorandum on the FDA website. You will see on page 1 that this is an application for a Biologics Licence for a product called JYNNEOS made by applicant Bavarian Nordic A/S for licensing of smallpox vaccines with “at Least 0.5 x 10^8 Infectious Units of Modified Vaccinia Ankara-BN (MVA-BN)”. Vaccinia virus is the well-known cow pox virus often used in (recombinant) vaccines. Vaccinia is also known to cause “certain complications and/or vaccine adverse effects occasionally arise” and, according to the people who want to promote the products, ~1/M people die from inoculation using vaccinia. That was before the COVID-19 injections however, and no one has any way to predict if these products may be counter-indicated, for whatever reason.
There are many redactions in this 209 page document. Never really understood the redaction thing; I mean, besides to respect a clinical trial participant’s privacy, for example, why would you do it? I suppose that considering we are pre-super-surveillance state, privacy probably isn’t really what these people are trying to protect, is it?
Read the following paragraph closely. It’s on page 189. Now, first of all, how is it that half of the world population probably knows what a frikkin’ troponin is in the first place? Could it be because of the frequency of reporting of massive elevations of troponins in clinical myocarditis cases diagnosed in temporal proximity to COVID-19 injections? Second of all, did the ‘applicant’ actually continue monitoring cardiac events as part of the post-marketing pharmacovigilance plan as recommended by the reviewer?
As you saw in my Twitter post, it is recommended in the presentation given by Brett Peterson in “Clinical Guidance for the use of JYNNEOS”, that people get counseled on the potential risk of myopericarditis given ‘its uncertain aetiology associated with replication-competent monkeypox vaccines’. And what about the potential risk of you’ve sustained heart damage from the COVID-19 shots?
There is at least one woman in the POX-MVA-005 trial with ongoing Cardiomyopathy that is ‘Possibly related’ in a very young woman who participated in the trial. Healthy, 28 years old. There’s also a 30 year-old male with myocardial infarction that was ‘Possibly related’ as well, that they claim has since ‘resolved’. By resolved, do you mean he died? I seriously have to ask.
I don’t expect you to read the whole page that follows here, but you should. ACAM2000 is a really unpleasant version of a smallpox vaccine. Basically. Here it is reported that 18.4% of participants in the trials involving POX-MVA-008 and POX-MVA-011 (both Bavarian Nordic Smallpox vaccines) showed post-vaccination elevation of troponin regardless of health status prior.
N.B. When I went to find our more about the POX-MVA-011 trial, I found this consent form document that describes the study where 2/3 of the study groups were meant to be HIV-infected people (18-55). Now, this is a study to compare HIV and non-HIV groups, and I do value the scientific question in here since I was an HIV-researcher, but this weird link between HIV and everything related to ‘public health’ lately strikes me as, re-occurring. I know I sound ‘paranoid’ but if they’re really out to get you, it ain’t paranoia, is it?
The purpose of this research study is to compare the safety and tolerability of a new investigational smallpox vaccine called MVA-BN in HIV infected subjects to healthy (HIV) negative subjects. In addition the induction of an immune response (the body's defense system) against smallpox in subjects with HIV
infection will be compared to healthy subjects.
Here are some more details outlined in the document as to what some of the participants experienced and reported.
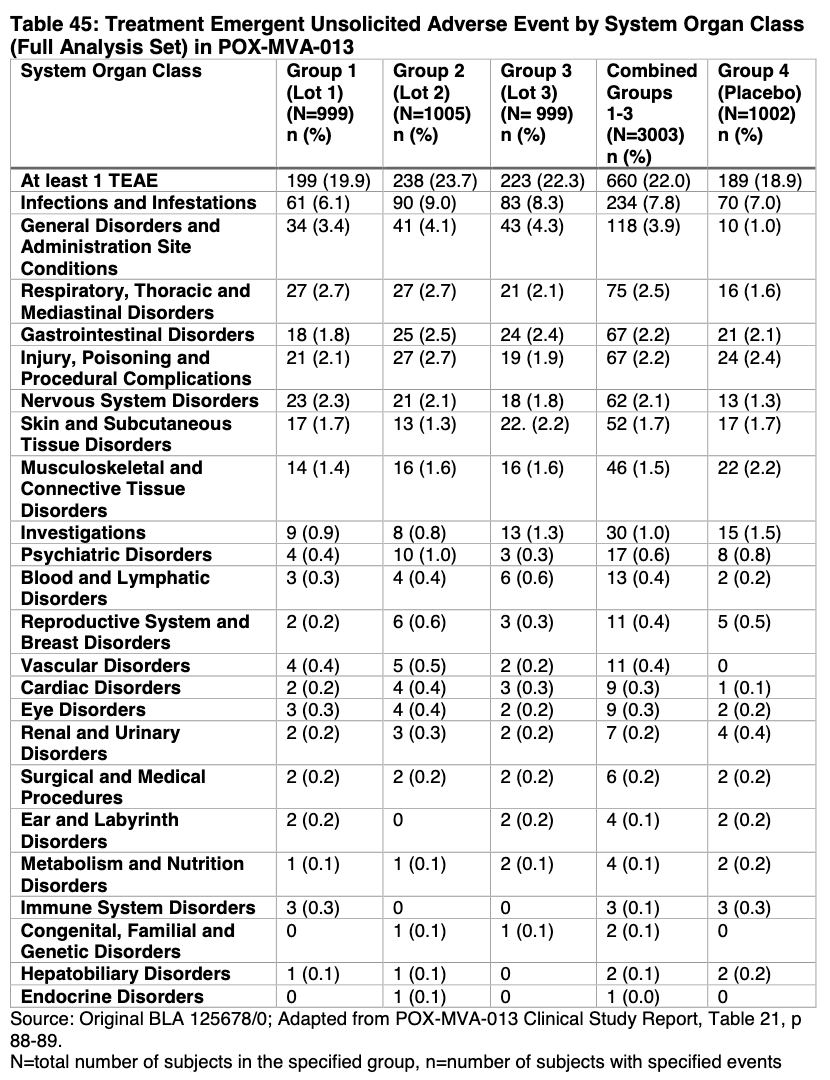
That’s all I have to report on cardiac stuff. I want to move on to foetal stuff. Below is a general table reporting “Treatment Emergent Unsolicited Adverse Event by System Organ Class in POX-MVA-013”. The thing that struck me here are the high rates of infections and infestations in all study groups. What’s with that? Are we talking Shingles here? Scabies? What? Since 9% of people in one of the study groups (Group 2) may have suffered a Shingles outbreak, for example, then that’s worth reporting on. Shingles is serious, contagious and horribly painful. And highly associated with the COVID-19 shots. Ahem.
You’ll also notice on this chart that they reported on Reproductive System and Breast Disorders. That’s good. They checked, at least. The numbers don’t look too outrageous (0.4%). But… Why did they redact the number of days since last vaccine for the Fetal death report here? And what do they mean by ‘Resolved’?
And they did it in every spontaneous abortion case and foetal death case but for no other Severe Adverse Event (SAE). Why? I mean, they reported on the convulsion episode that was reported on the 5th day after the last vaccine. Maybe it was because the young man was known to have had seizures in childhood?
My guess would be that they didn’t want anyone to know the short temporal distance between the injection and abortion because it might make someone think that the shots were causing abortions. Just a guess though.
Just one last share. There were occurrences of adverse events in 97.7% of the participants in the ACAM2000 study - do not let anyone you love take that shot. The ACAM2000 product is FDA approved, believe it or not, and does come with some serious warnings.
1/3 of all healthy participants experienced an adverse event in the context of the MVA-BN shots. These represent the Modified Vaccinia Ankara (MVA) products made by Bavarian Nordic (BN).
Smallpox vaccine development is now in its second generation. First-generation vaccines were derived from calf-lymph, and include Dryvax, APVS, Lancy-vaxina and Lister. Second-generation vaccines are cell-cultured and include ACAM2000 and CCSV.
Both Dryvax and [ACAM2000] come from the New York City Board of Health strain of vaccinia. Dryvax was grown on calf skin and then freeze-dried for storage. Dryvax was first licensed by the FDA in 1931; however, it is no longer manufactured. ACAM2000 is a second generation smallpox vaccine. It comes from a clone of Dryvax which is purified and produced using modern cell culture technology.
I suppose that JYNNEOS is the next generation?

My bottom line here is this: when they start insisting that we are in another emergency and that everyone needs to comply and get injected with this JYNNEOS stuff because otherwise you’ll die or get black-bagged, I will insist harder that we have no goddamned idea what the effects of chasing the COVID-19 novel tech with this product will be. It is clearly written down, even by the FDA, that people with heart issues/myocarditis should consider it risky to take the smallpox injections, so if one experienced an adverse event involving heart injury from the COVID-19 shots, then they should be categorically denied (out of public health safety concerns, of course!) access to the smallpox/monkeypox shot since they have been shown to independently be associated with myocarditis in PREVIOUSLY HEALTHY PEOPLE.
In lieu of this, there are solutions. The main one still being - keeping your Vitamin D and Zinc levels up to standard (not be deficient).
Enjoy the moneypox fear circus.
https://en.wikipedia.org/wiki/ACAM2000













I can’t believe I am watching all of this happen in real time. If it was a movie I would walk out and say that it would never happen in real life.
I was going to write about JYNNEOS but I'll post here instead.
Obviously this is just a coincidence but has anyone watched the UK Channel 4 series, Utopia?
I highly recommend it.
It's about a group of people who think global overpopulation needs sorting. They develop a serum to sterilise 90-95% of the planet. It is split in two parts to avoid detection. The first part is in industrially farmed corn and the other part in a vaccine produced after the population is scared by Russian flu. Only when the two components combine are the effects triggered but separately undetectable.
The name of the vaccine is Janus. Sound a bit like JYNNEOS?